Veterinary telemedicine bill prompts split testimony over access and federal prescribing rules
February 19, 2026 | Legislative Sessions, Washington
This article was created by AI summarizing key points discussed. AI makes mistakes, so for full details and context, please refer to the video of the full meeting. Please report any errors so we can fix them. Report an error »
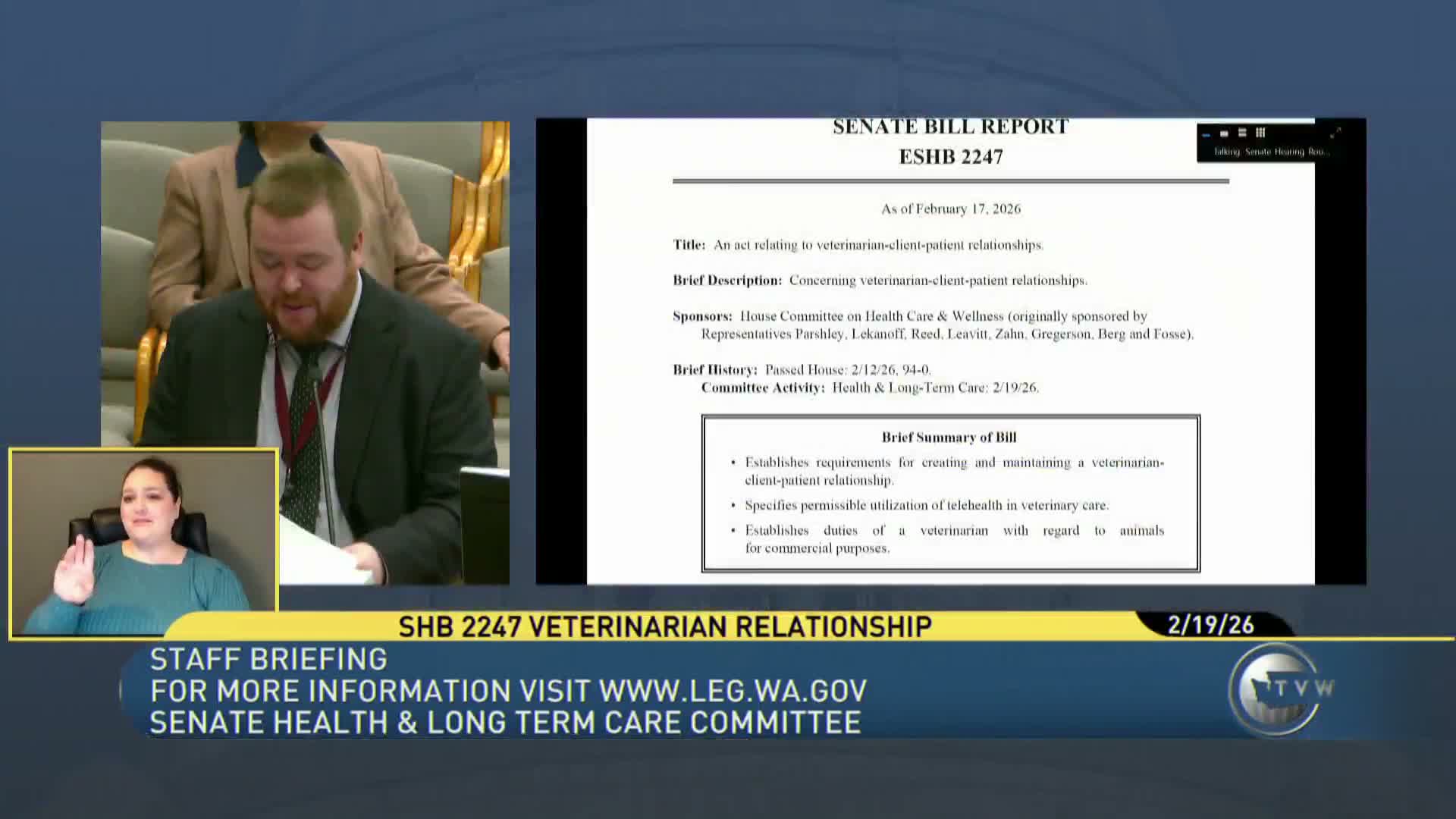
House Bill 2,247, which would modify the veterinary client‑patient relationship to permit certain telemedicine interactions, produced extensive testimony on Feb. 19 as the Senate committee reopened hearings.
Proponents — including animal‑welfare organizations, veterinarians who use telemedicine, and telemedicine advocacy groups — described telemedicine as a proven tool in nine other states that helps reach clients who lack transportation, live in under‑resourced areas, or face long waits for appointments. Proponent witnesses said the bill includes guardrails (licensure in Washington, recordkeeping, limits on commercial operations) and noted federal guidance that allows state authorities to set VCPR rules for on‑label use.
The state veterinarian and representatives of the Washington State Veterinary Medical Association urged caution in the bill’s current form. State Veterinarian Amber Ittle said the bill as drafted conflicts with FDA and USDA requirements that an in‑person exam be performed to establish a valid VCPR before prescribing extra‑label medications; she highlighted public‑health and food‑safety risks including zoonotic disease surveillance and drug‑residue concerns. Veterinary board representatives and the association asked the committee to reinstate language limiting extra‑label prescribing or otherwise align the bill with federal mandates.
Witnesses on both sides suggested amendments: proponents urged passage with guardrails to expand access, while critics proposed adding or restoring explicit on‑label/extra‑label language to avoid conflict with federal law. The committee heard dozens of proponent witnesses and a smaller number of opponents; testimony closed and the committee adjourned for the day without taking final action on HB 2,247.
Proponents — including animal‑welfare organizations, veterinarians who use telemedicine, and telemedicine advocacy groups — described telemedicine as a proven tool in nine other states that helps reach clients who lack transportation, live in under‑resourced areas, or face long waits for appointments. Proponent witnesses said the bill includes guardrails (licensure in Washington, recordkeeping, limits on commercial operations) and noted federal guidance that allows state authorities to set VCPR rules for on‑label use.
The state veterinarian and representatives of the Washington State Veterinary Medical Association urged caution in the bill’s current form. State Veterinarian Amber Ittle said the bill as drafted conflicts with FDA and USDA requirements that an in‑person exam be performed to establish a valid VCPR before prescribing extra‑label medications; she highlighted public‑health and food‑safety risks including zoonotic disease surveillance and drug‑residue concerns. Veterinary board representatives and the association asked the committee to reinstate language limiting extra‑label prescribing or otherwise align the bill with federal mandates.
Witnesses on both sides suggested amendments: proponents urged passage with guardrails to expand access, while critics proposed adding or restoring explicit on‑label/extra‑label language to avoid conflict with federal law. The committee heard dozens of proponent witnesses and a smaller number of opponents; testimony closed and the committee adjourned for the day without taking final action on HB 2,247.
View the Full Meeting & All Its Details
This article offers just a summary. Unlock complete video, transcripts, and insights as a Founder Member.
✓
Watch full, unedited meeting videos
✓
Search every word spoken in unlimited transcripts
✓
AI summaries & real-time alerts (all government levels)
✓
Permanent access to expanding government content
30-day money-back guarantee
